Slideshow Items
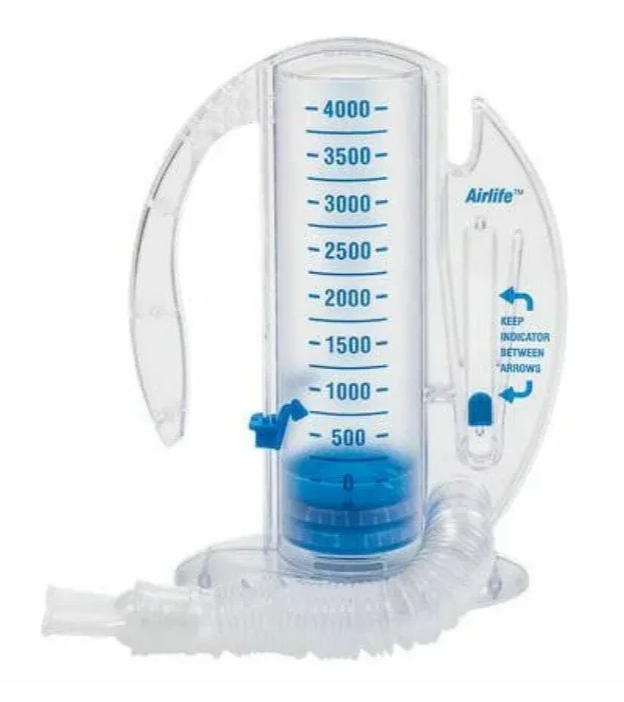
Latex-free 4,000 mL volumetric incentive spirometer with a dual-sided calibration scale, adjustable patient goal indicator, and one-way valve, guiding slow deep-breathing exercises for post-operative lung expansion and pulmonary recovery.
Loading…
The AirLife 4,000 mL volumetric incentive spirometer serves two structurally distinct functions that are both essential to effective post-operative and respiratory recovery, and that a single device must satisfy simultaneously. The first function is lung expansion feedback: the spirometer is the measurement and visualisation instrument that converts the patient's deep inhalation effort into a visible, quantifiable volume reading observable in real time during each breathing exercise. Patients recovering from surgery, prolonged bed rest, or respiratory illness tend toward shallow, low-volume breathing — a natural protective response to incisional pain, reduced respiratory muscle activity, or general debilitation. Shallow breathing does not fully inflate the distal alveoli, allowing these alveolar units to progressively collapse — a condition called atelectasis — that reduces the functional lung volume available for gas exchange and creates the conditions for post-operative pneumonia. Incentive spirometry addresses this tendency by providing the patient with a concrete, visible volume target for each breath: the graduated calibration scale shows exactly how much air their inhalation has drawn through the device, allowing them to compare each breath against a prescribed target and observe whether their effort is reaching the therapeutically effective expansion level. Without this feedback, the patient has no reliable way to judge breathing depth — the subjective sense of a deep breath is a consistently poor indicator of actual lung expansion volume, and patients routinely underestimate the additional effort required to reach therapeutically effective tidal volumes during post-operative recovery.
The second function the spirometer must satisfy simultaneously is atelectasis prevention — the clinical purpose for which incentive spirometry is prescribed. Post-operative atelectasis is particularly common following thoracic and abdominal surgery, where incisional pain limits voluntary inhalation depth, the supine position reduces gravitational assistance to diaphragmatic descent, and the residual effects of anaesthetic agents reduce respiratory drive. The incentive spirometer's deep inhalation coaching function addresses atelectasis by directing the patient to take a slow, sustained maximum inhalation that generates the sustained distending pressure within the alveoli that re-opens collapsed alveolar units and maintains them open through the breath-hold period following peak volume. A rapid, forceful inhalation — the instinctive deep breath patients take without coaching — generates a similar peak volume but does not provide the sustained alveolar pressure duration that opens collapsed alveoli as effectively as a slow, sustained effort. The spirometer's design rewards the sustained technique by requiring the patient to draw the volume indicator upward through the full calibrated range with a controlled inhalation rather than a sharp effort.
The adjustable patient goal indicator of the 4,000 mL AirLife spirometer addresses the clinical requirement of setting and communicating an individualised volume target for each patient's breathing exercises. The target volume for incentive spirometry is not uniform across patients — it is based on the patient's predicted inspiratory capacity, which varies with height, age, sex, and baseline pulmonary function. A patient with a larger predicted inspiratory capacity requires a higher target volume to achieve adequate alveolar recruitment than a patient with a smaller baseline capacity. The adjustable goal indicator allows the clinician or respiratory therapist to mark the patient's specific target volume on the calibration scale at the point of device issue, giving the patient a clear, personalised reference point for every subsequent exercise session without requiring the clinician to be present for each session. This individualisation is clinically significant — a single standard target volume that is too low for the patient's capacity provides insufficient alveolar recruitment, while a target that is set too high may cause the patient to abandon the exercise as unachievable.
The 4,000 mL chamber capacity of this AirLife spirometer extends the effective volume range above the 2,500 mL standard model, accommodating the larger inspiratory capacities of taller adult patients and of patients who have progressed in their respiratory recovery to where their achievable inhalation volume has increased beyond the 2,500 mL ceiling. The dual-sided calibration scale provides volume markings readable from either side of the device, accommodating right-handed and left-handed patients without requiring the device to be rotated — a usability detail that matters during bed-based exercises where the patient's ability to reposition the device may be limited by IV lines, surgical drains, or physical discomfort.
✓ Post-operative respiratory recovery — incentive spirometry prescribed at regular intervals following thoracic, abdominal, or other major surgery where respiratory muscle restriction and anaesthetic effects increase atelectasis risk, with the 4,000 mL capacity accommodating the full adult patient range
✓ Taller and larger adult patients — 4,000 mL chamber capacity recommended for patients whose height and predicted inspiratory capacity would exceed the 2,500 mL ceiling of the standard model, ensuring the target volume can be prescribed and achieved without chamber limitation
✓ Recovery progression tracking — adjustable goal indicator allows the prescribed target volume to be progressively increased as the patient's achievable inhalation volume improves during the recovery period, tracking rehabilitation progress against a visible advancing target
✓ Post-operative pneumonia prevention — regular deep-breathing exercises with spirometer feedback supporting alveolar recruitment and secretion mobilisation that reduces post-operative pneumonia incidence in surgical recovery protocols
✓ Respiratory illness rehabilitation — deep-breathing programme support for patients recovering from pneumonia, pleural effusion, or other respiratory conditions where reduced functional lung volume requires coached deep-breathing exercises to restore alveolar ventilation
Carousel items
Testimonial items
Very welcoming and informative. We went in to rent a Walker for my mom to see if she would use it. They had no rentals left so he gave us a brand new one on rental. Highly recommend this company for all your ADL needs.
Tara Maye
The rating of this product is 5 out of 5
Fantastic service and experience, from delivery to pickup we could not have asked for anything more! We rented a hospital bed, and I do not believe you would get better service anywhere. Highly recommended!
Shawn Dillon
The rating of this product is 5 out of 5
Super friendly and very helpful! Delivered the wheelchair for me, special ordered other parts and took the time to show me how to install. I recommend!
Fiona Haines
The rating of this product is 5 out of 5
Can not thank the team at Med Supplies enough for their amazing service. We were in a tough spot till we got their help. Amazing service. Kind and respectful delivery. First class all the way. Thank you again.
Jon Beatty
The rating of this product is 5 out of 5
Ordered the chair on Sunday and it arrived Monday morning. Spoke to customer service to follow up on delivery times. It was already on my front door. Excellent and helpful staff. The product is sturdy and of good quality. Thank you for your help.
H D
The rating of this product is 5 out of 5
Excellent experience - website faithfully represented what was in stock (which hasn't always been my experience with other vendors sadly), and local shipping was really fast - ordered on the weekend, received it on Monday in my case. Thank you for being
Jason Hudson
The rating of this product is 5 out of 5
0/0
You cart is currently empty
 780 409-1509
780 409-1509 Get directions
Get directions