Slideshow Items
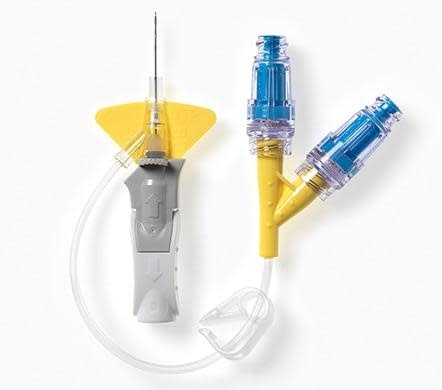
BD Nexiva Closed IV Catheter System (Dual Port, 24G x 0.75", 19 mm) is a micro-gauge pre-assembled closed system with Vialon biomaterial and BD Q-Syte connector for fragile veins. Reduces blood exposure and vascular trauma during IV therapy.
Loading…
The 24 Gauge configuration of the Nexiva Closed IV Catheter System is designed for the patient populations and vein conditions where a larger gauge catheter would cause unnecessary trauma or be impossible to place: paediatric patients, elderly patients with fragile and thin-walled veins, oncology patients with chemotherapy-damaged vasculature, and any clinical situation where minimising vascular trauma is the primary insertion consideration. At 24G, the catheter is at the smaller end of the standard peripheral IV gauge range — appropriate for slow infusion therapies, medication delivery, and fluid maintenance where flow rates above approximately 18 mL/min are not required. The Vialon™ biomaterial softens and becomes more flexible in the vein after insertion, reducing the mechanical irritation to delicate vessel walls that is the primary source of phlebitis and early catheter failure in fragile veins. The closed system design keeps the fluid pathway sealed during insertion, significantly reducing blood exposure for the clinician. Blood control technology helps contain blood at the hub during insertion. The BD Q-Syte™ needle-free connector and dual port design allow medication delivery and flushing without disconnecting the line. The integrated stabilisation platform minimises catheter movement to reduce dislodgement and infiltration risk. Yellow colour-coded hub for immediate 24G identification.
Paediatric patients requiring peripheral IV access where a smaller 24G catheter minimises insertion trauma and is appropriate for child-sized veins
Geriatric and elderly patients with fragile, thin-walled, or rolling veins where a 24G catheter reduces vascular trauma and improves catheter dwell time
Oncology patients with chemotherapy-damaged vasculature requiring the smallest gauge appropriate for their infusion therapy
Patients requiring slow infusion therapy — maintenance fluids, antibiotic infusions, or parenteral nutrition — where 24G flow rates are clinically adequate
Emergency and urgent care settings where IV access must be established in difficult veins with minimal trauma and a first-stick success priority
Clinical staff with occupational blood exposure risk reduction requirements where the closed system design provides additional protection during IV insertion
Carousel items
Testimonial items
Very welcoming and informative. We went in to rent a Walker for my mom to see if she would use it. They had no rentals left so he gave us a brand new one on rental. Highly recommend this company for all your ADL needs.
Tara Maye
The rating of this product is 5 out of 5
Fantastic service and experience, from delivery to pickup we could not have asked for anything more! We rented a hospital bed, and I do not believe you would get better service anywhere. Highly recommended!
Shawn Dillon
The rating of this product is 5 out of 5
Super friendly and very helpful! Delivered the wheelchair for me, special ordered other parts and took the time to show me how to install. I recommend!
Fiona Haines
The rating of this product is 5 out of 5
Can not thank the team at Med Supplies enough for their amazing service. We were in a tough spot till we got their help. Amazing service. Kind and respectful delivery. First class all the way. Thank you again.
Jon Beatty
The rating of this product is 5 out of 5
Ordered the chair on Sunday and it arrived Monday morning. Spoke to customer service to follow up on delivery times. It was already on my front door. Excellent and helpful staff. The product is sturdy and of good quality. Thank you for your help.
H D
The rating of this product is 5 out of 5
Excellent experience - website faithfully represented what was in stock (which hasn't always been my experience with other vendors sadly), and local shipping was really fast - ordered on the weekend, received it on Monday in my case. Thank you for being
Jason Hudson
The rating of this product is 5 out of 5
0/0
You cart is currently empty
 780 409-1509
780 409-1509 Get directions
Get directions