Slideshow Items
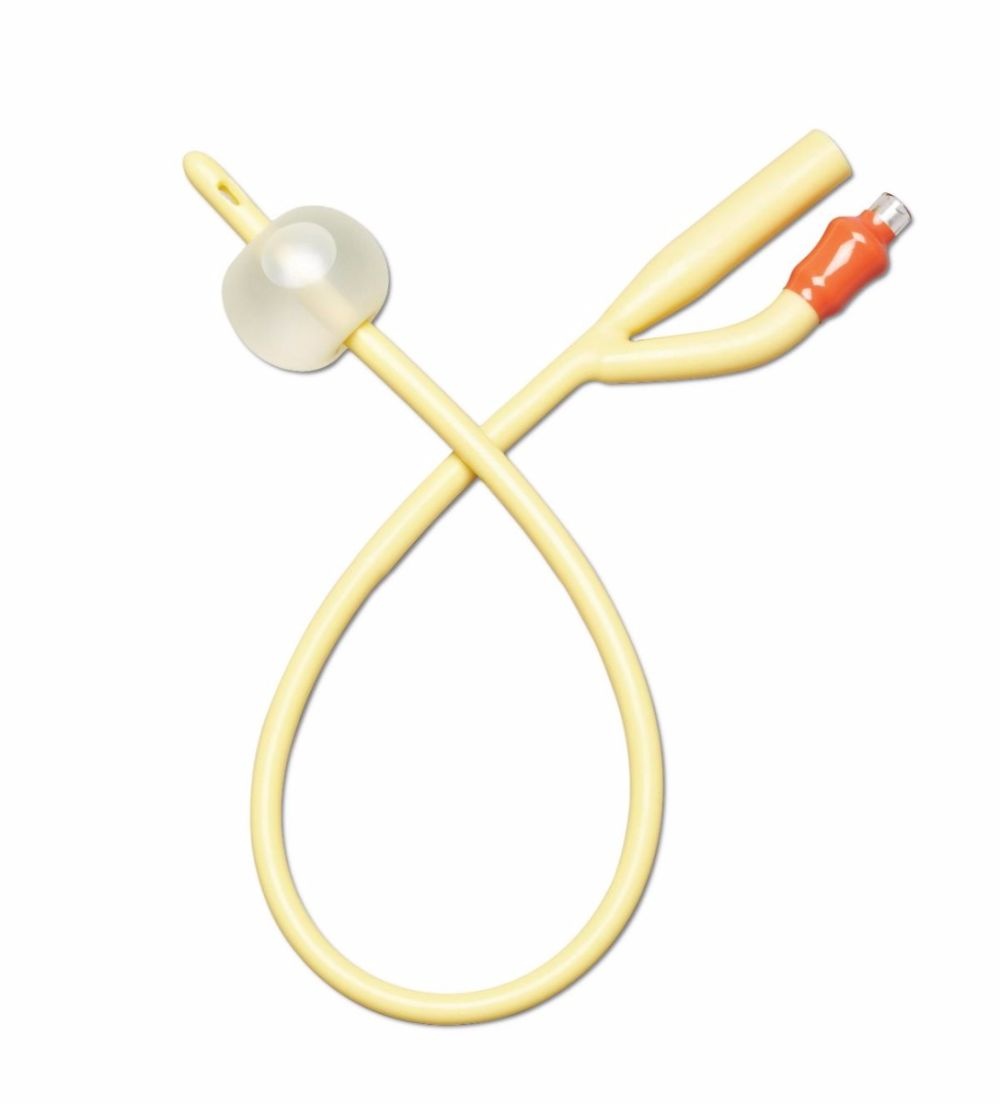
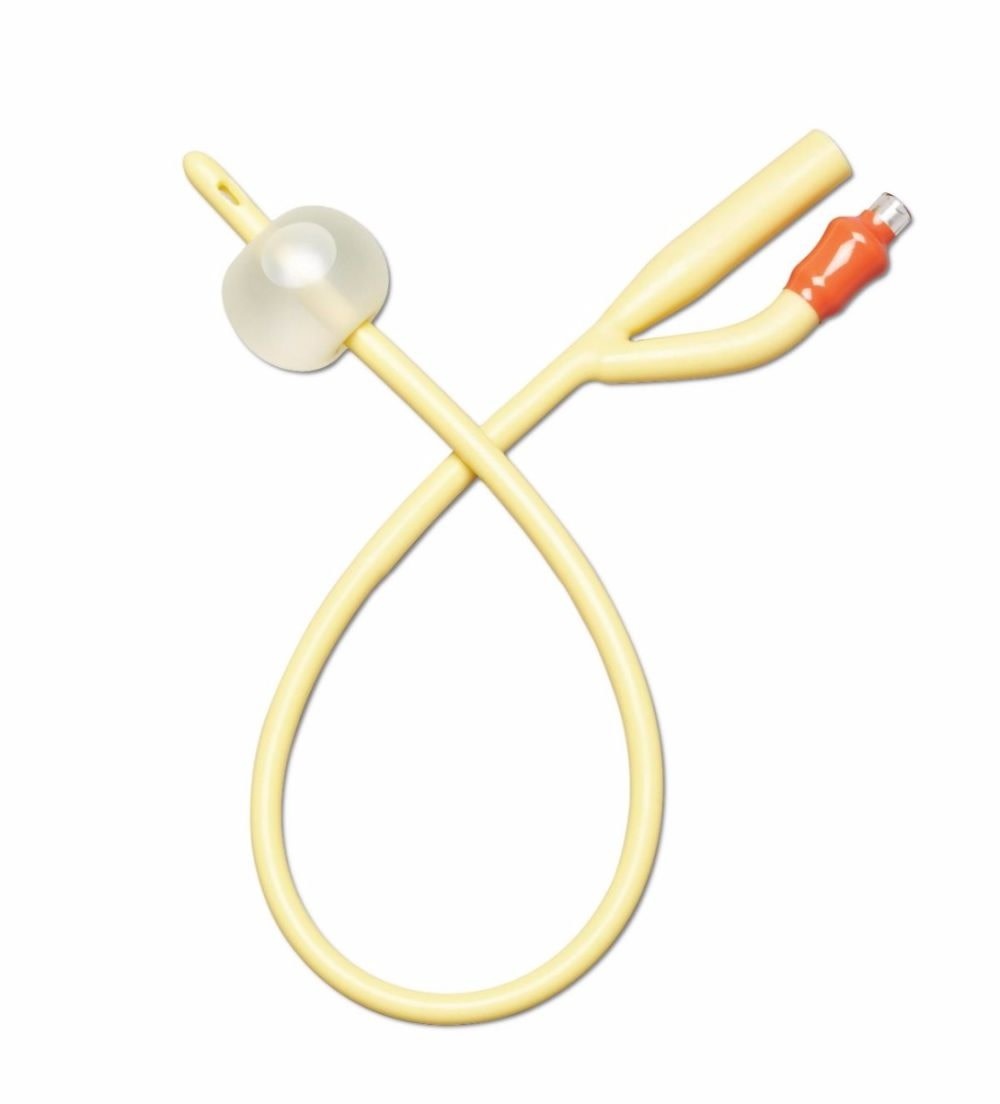
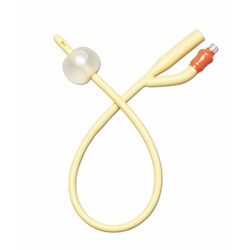
2-way Foley features silicone elastomer-coated shaft and polished eyelets for gentle insertion and reliable drainage. 10 mL balloon provides secure retention. Color-coded funnel simplifies 16 Fr size identification.
Loading…
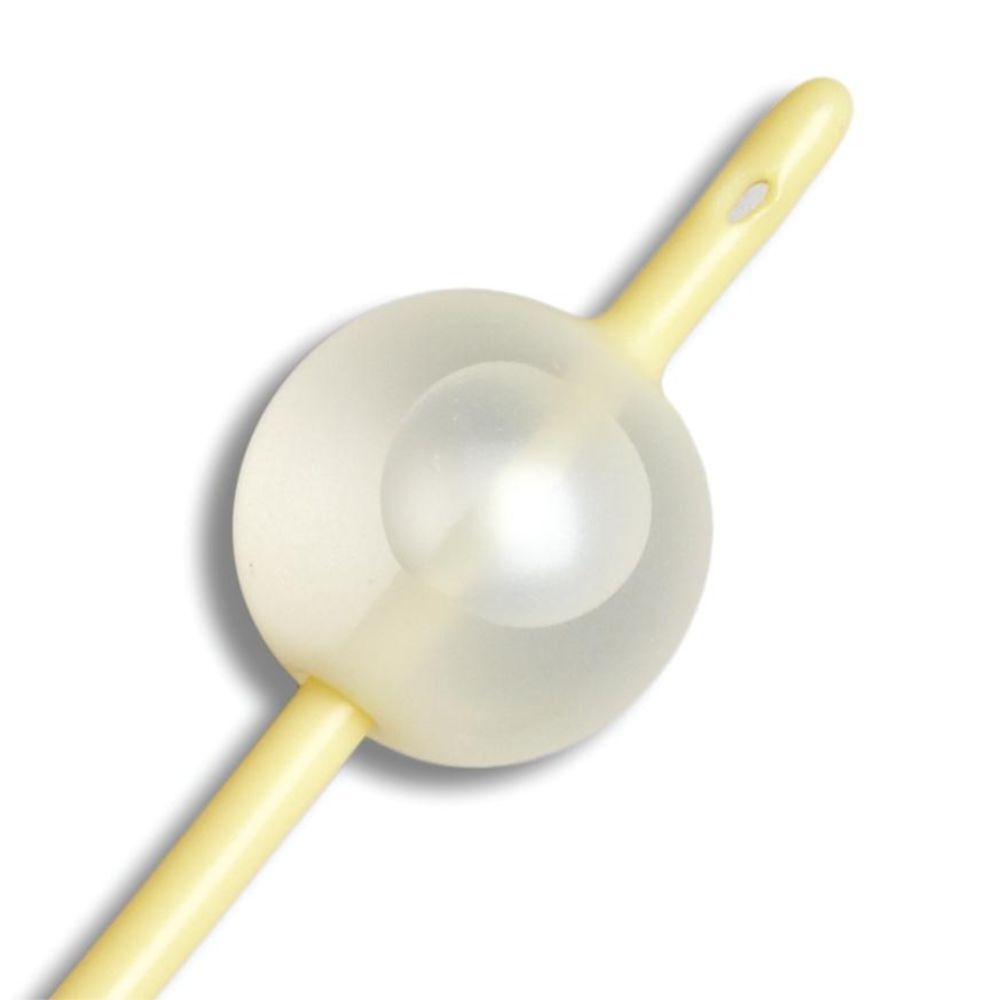
This catheter is built for indwelling urinary drainage — catheter placement that remains in the bladder for continuous drainage over hours to days, rather than the in-and-out single-use insertion of an intermittent catheter routine. Indwelling Foley catheterization is the standard approach in post-surgical recovery, ICU and ward care, acute urinary retention, hospice and palliative care, and long-term care users who cannot self-catheterize or void normally. Four structural specifications define this SKU's clinical position: the 2-way design, the silicone elastomer coating on a latex-free shaft, the sterile packaging, and the 10cc retention balloon. The 2-way (or "two-channel") design is the basic Foley configuration — one lumen drains urine from the bladder through the catheter shaft to an external drainage bag, and a second smaller lumen carries sterile water to the retention balloon at the catheter tip. The 2-way design suits routine drainage; 3-way Foleys add a third lumen for continuous bladder irrigation (used after urological surgery or in specific clot-management scenarios), which is not the use case for this SKU. The silicone elastomer coating on a latex-free shaft is the material specification that positions this Foley distinctly from silicone-coated latex alternatives. Silicone-coated latex catheters (such as the Dover Kenguard line) use a latex core for pliability with a silicone surface layer — but the latex core still presents allergy risk to latex-sensitive patients and the silicone coating reduces rather than eliminates latex contact. A latex-free silicone elastomer catheter eliminates the latex allergy concern entirely — appropriate for patients with confirmed latex allergy or sensitivity, and a common default selection in clinical settings where latex sensitivity status is unknown or where facility policy requires latex-free defaults across all catheter inventory. The silicone elastomer coating reduces surface friction during insertion and supports a smoother catheter shaft profile compared with uncoated alternatives. The sterile packaging is the clinical-procedure standard — required for sterile-technique insertion in clinical and facility CAUTI prevention protocols where the catheter must enter the sterile field through an intact sterile barrier. The 10cc retention balloon is the bladder-anchoring mechanism: after insertion the balloon is inflated with sterile water per the manufacturer's specified volume (verify against current clinical protocol — 10cc nominally indicates the balloon's intended inflation volume, but facility CAUTI protocols may specify different volumes), and the inflated balloon sits inside the bladder against the internal urethral opening, preventing the catheter from being pulled or dislodged during patient movement, transfers, or drainage bag handling. The 10cc balloon size suits routine adult indwelling use — larger 30cc balloons exist for specific haemostasis applications after urological surgery. The 16Fr French size is a common adult indwelling calibre — slightly larger than the 14Fr also commonly used, supporting marginally higher drainage flow in scenarios where output viscosity or volume favours a larger lumen. Medline is a large US-origin medical supply manufacturer with a long-standing position in catheter and drainage system design. Use only under healthcare professional direction — Foley catheterization is a clinical procedure with specific insertion, securement, and surveillance protocols that govern catheter-associated urinary tract infection (CAUTI) risk management.
✔ Medline indwelling Foley urinary catheter
✔ Construction: Silicone elastomer-coated, latex-free
✔ Catheter size: 16Fr — common adult indwelling calibre
✔ Balloon size: 10cc / 10 ml (verify inflation volume against clinical protocol)
✔ Design: 2-way (drainage lumen + balloon inflation lumen)
✔ Sterile packaging for clinical sterile-technique insertion
✔ Latex-free — suitable for latex-allergic patients
✔ Silicone elastomer coating for reduced insertion friction
✔ Retention balloon system for bladder anchoring
✔ Drainage lumen and balloon inflation lumen integrated in shaft
✔ Useful for clinical care, facility care, and caregiver-assisted routines under professional direction
✔ 2-way Foley design supports continuous indwelling urinary drainage with bladder retention via inflated balloon
✔ Latex-free silicone elastomer construction eliminates latex allergy risk — appropriate for confirmed latex-allergic patients and latex-default facility inventory
✔ Silicone elastomer coating reduces surface friction during insertion compared with uncoated alternatives
✔ 10cc balloon anchors the catheter in the bladder — prevents dislodgement during patient movement, transfers, or drainage bag handling
✔ 16Fr calibre supports clinically reasonable drainage flow at a commonly used adult size — slightly larger than 14Fr for higher-viscosity or higher-volume output scenarios
✔ Sterile packaging supports clinical and facility sterile-technique insertion per CAUTI prevention protocols
✔ Useful for post-surgical recovery, acute retention, ICU, ward, hospice, and long-term care indwelling drainage scenarios
✔ Latex-free default supports facility inventory simplification — single catheter type suits all patients regardless of latex sensitivity status
✔ Post-surgical and post-procedural indwelling urinary drainage in clinical settings
✔ Acute urinary retention requiring continuous bladder drainage
✔ ICU, ward, hospice, and long-term care indwelling Foley management
✔ Confirmed latex-allergic patients requiring non-latex Foley alternatives
✔ Facility policy requiring latex-free defaults across all catheter inventory
✔ Routine 2-way Foley scenarios — not for continuous bladder irrigation (3-way Foley required)
✔ Higher-viscosity or higher-volume output scenarios favouring 16Fr over 14Fr calibre
✔ Clinical and facility supply for routine Foley resupply at the 16Fr adult calibre
Use only under healthcare professional direction. Confirm the prescribed catheter size (16Fr), balloon volume, Foley design (2-way vs 3-way), and latex-free requirement match the clinical order. Use sterile insertion technique per facility CAUTI prevention protocol — the sterile packaging supports sterile-field maintenance during insertion. Lubricate the catheter as directed and insert per prescribed clinical technique — do not force against resistance. Confirm urine return through the drainage lumen before inflating the balloon to verify intravesical placement. Inflate the balloon with sterile water per current clinical guidance and manufacturer specification — verify the inflation volume against the catheter labelling and current CAUTI prevention protocols. Secure the catheter to the patient's thigh or abdomen per facility securement protocol to prevent traction. Connect to a closed drainage bag system — maintain the closed system between scheduled bag changes. Position the drainage bag below bladder level at all times. Document insertion, indications, and ongoing daily catheter necessity review per CAUTI prevention protocol. Replace the catheter according to clinical schedule or sooner if obstruction, leakage, or signs of infection occur. Remove the catheter as soon as clinically appropriate — duration of indwelling catheterization is a primary CAUTI risk driver. Dispose of used catheters following local clinical waste guidance. Store unused catheters in original sterile packaging at room temperature away from heat and direct sunlight.
Carousel items
Testimonial items
Very welcoming and informative. We went in to rent a Walker for my mom to see if she would use it. They had no rentals left so he gave us a brand new one on rental. Highly recommend this company for all your ADL needs.
Tara Maye
The rating of this product is 5 out of 5
Fantastic service and experience, from delivery to pickup we could not have asked for anything more! We rented a hospital bed, and I do not believe you would get better service anywhere. Highly recommended!
Shawn Dillon
The rating of this product is 5 out of 5
Super friendly and very helpful! Delivered the wheelchair for me, special ordered other parts and took the time to show me how to install. I recommend!
Fiona Haines
The rating of this product is 5 out of 5
Can not thank the team at Med Supplies enough for their amazing service. We were in a tough spot till we got their help. Amazing service. Kind and respectful delivery. First class all the way. Thank you again.
Jon Beatty
The rating of this product is 5 out of 5
Ordered the chair on Sunday and it arrived Monday morning. Spoke to customer service to follow up on delivery times. It was already on my front door. Excellent and helpful staff. The product is sturdy and of good quality. Thank you for your help.
H D
The rating of this product is 5 out of 5
Excellent experience - website faithfully represented what was in stock (which hasn't always been my experience with other vendors sadly), and local shipping was really fast - ordered on the weekend, received it on Monday in my case. Thank you for being
Jason Hudson
The rating of this product is 5 out of 5
0/0
Recently view items
You cart is currently empty
 780 409-1509
780 409-1509 Get directions
Get directions